Pilot Study on the Comparison of Seven Commonly Used Over-the-Counter Pulse Oximeter Finger Devices
DOI:
https://doi.org/10.70172/pjcd.v23i1.10201Keywords:
pulse oximeter, precision, accuracyAbstract
Background: Over-the-counter pulse oximeters are commonly used in patient care but have yet to be evaluated for their accuracy and precision. The objective of the study was to compare the accuracy of seven over-the-counter pulse oximeter finger devices in measuring oxygen saturation (SpO2), with arterial blood gas oxygen saturation (SaO2) as the reference value.
Methodology: This was a cross-sectional study from June to November 2023. Pulse oximeter readings of participants aged 18 and above were compared with their SaO2. Precision was calculated using mean bias and standard deviation while accuracy was assessed through average root mean square error (ARMS). Subgroup analysis of patients with hypoxemia (SaO2 <95%) was done.
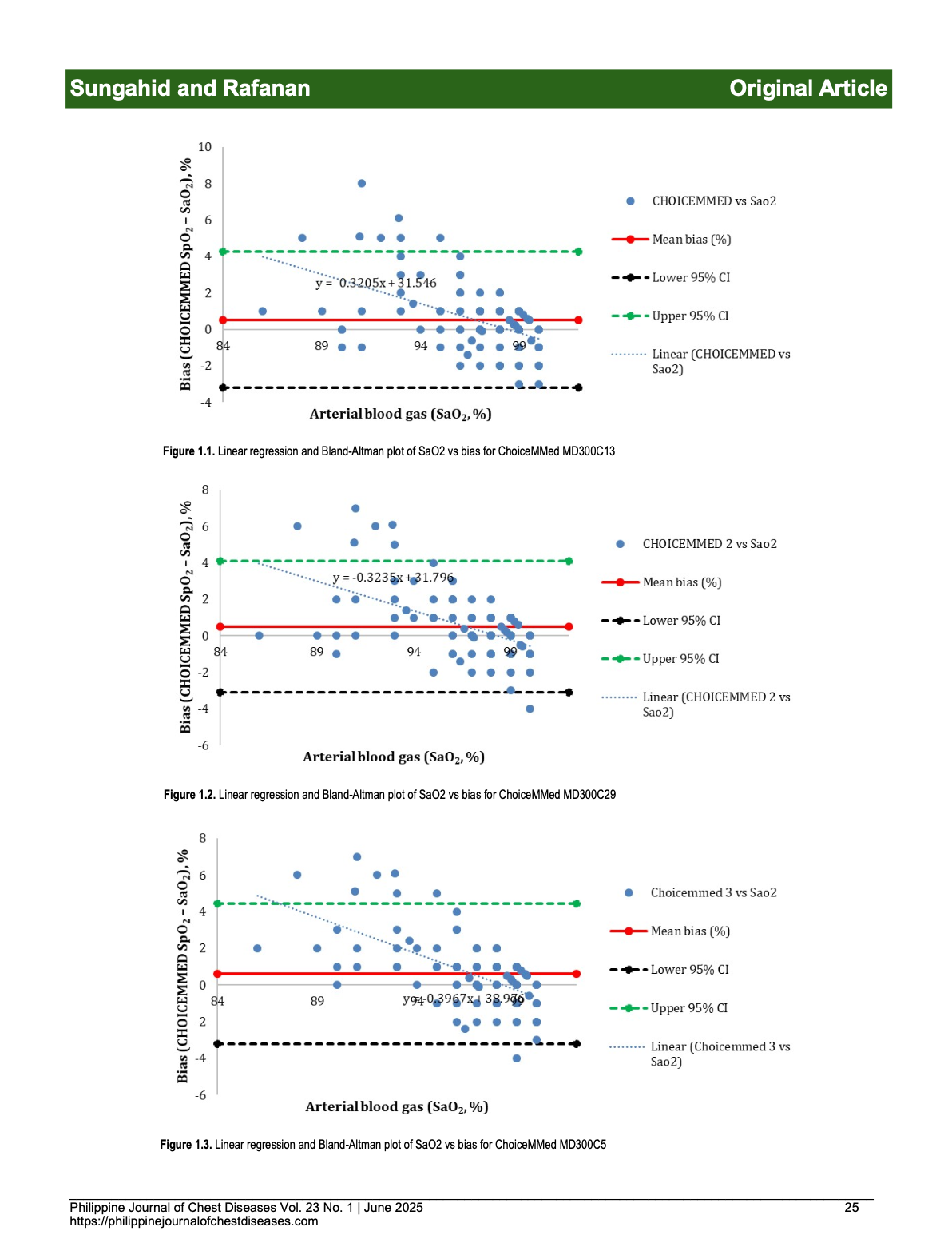
Results: The study included 107 participants. Among the devices, ChoiceMMed MD300C29 had the lowest standard deviation (±1.84) and Yongrow showed the lowest mean bias (0.21), indicating higher precision among the devices studied. ChoiceMMed MD300C29 was the most accurate based on the ARMS score (1.89%). In contrast, IMDK had the highest mean bias (-0.90) and the largest standard deviation (±2.47), indicating lower precision. Hypoxemic subgroup analysis revealed significant biases, lower precision, and lower accuracy for all devices.
Conclusion: ChoiceMMed MD300C29 and Yongrow demonstrate superior precision, with the former emerging as the most accurate. IMDK lags with the lowest precision and accuracy. Subgroup analysis shows less precision and accuracy of readings for hypoxemic patients, cautioning against the reliance on over-the-counter pulse oximeters for accurate SpO2 measurements. For enhanced accuracy, the study recommends the use of devices with lower mean bias, standard deviation, and ARMS values.
-----
This article has supplementary materials which can be accessed here.
References
U.S. Food and Drug Administration. Performance evaluation of pulse oximeters taking into consideration skin pigmentation, race and ethnicity. Prepared for the February 2, 2024, meeting of the Anesthesiology and Respiratory Therapy Devices Panel of the Medical Devices Advisory Committee. Center for Devices and Radiological Health (CDRH), U.S. Food and Drug Administration; 2024:1,3,30. Accessed April 28, 2025. https://www.fda.gov/news-events/press-announcements/fda-proposes-updated-recommendations-help-improve-performance-pulse-oximeters-across-skin-tones
International Organization for Standardization. ISO 9919:2005(E) Medical electrical equipment—Particular requirements for the basic safety and essential performance of pulse oximeter equipment for medical use. Geneva: ISO; 2005: viii.
Pilcher J, Ploen L, McKinstry S, Bardsley G, Chien J, Howard L, et al. A multicentre prospective observational study comparing arterial blood gas values to those obtained by pulse oximeters used in adult patients attending Australian and New Zealand hospitals. BMC Pulm Med. 2020;20(1):2. https://doi.org/10.1186/s12890-019-1007-3 DOI: https://doi.org/10.1186/s12890-019-1007-3
Harskamp RE, van der Laan AM, van Goor H, et al. Performance of popular pulse oximeters compared with simultaneous arterial oxygen saturation or clinical-grade pulse oximetry: A cross-sectional validation study in intensive care patients. BMJ Open Respir Res. 2021;8(1):e000939. https://doi.org/10.1136/bmjresp-2021-000939 DOI: https://doi.org/10.1136/bmjresp-2021-000939
Schrading WA, McCafferty B, Grove J, Page DB. Portable, consumer-grade pulse oximeters are accurate for home and medical use: implications for use in the COVID-19 pandemic and other resource-limited environments. JACEP Open. 2020;1(6):1450-1458. https://doi.org/10.1002/emp2.12292 DOI: https://doi.org/10.1002/emp2.12292
U.S. Food and Drug Administration. Pulse oximeter accuracy and limitations: FDA safety communication. 2022. Accessed April 28, 2025. https://www.fda.gov/media/162709/download
Shi C, Goodall M, Dumville J, Hill J, Norman G, Hamer O, et al. The accuracy of pulse oximetry in measuring oxygen saturation by levels of skin pigmentation: a systematic review and meta-analysis. BMC Med. 2022;20(1):267. https://doi.org/10.1186/s12916-022-02452-8 DOI: https://doi.org/10.1186/s12916-022-02452-8
Herbst A, Goel S, Beane A, Brotherton BJ, Dula D, Ely EW, et al. Oxygen saturation targets for adults with acute hypoxemia in low and lower-middle income countries: a scoping review with analysis of contextual factors. Front Med (Lausanne). 2023;10:1148334. https://doi.org/10.3389/fmed.2023.1148334 DOI: https://doi.org/10.3389/fmed.2023.1148334
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Sara Kristel P. Sungahid, MD, Albert L. Rafanan, MD

This work is licensed under a Creative Commons Attribution 4.0 International License.







